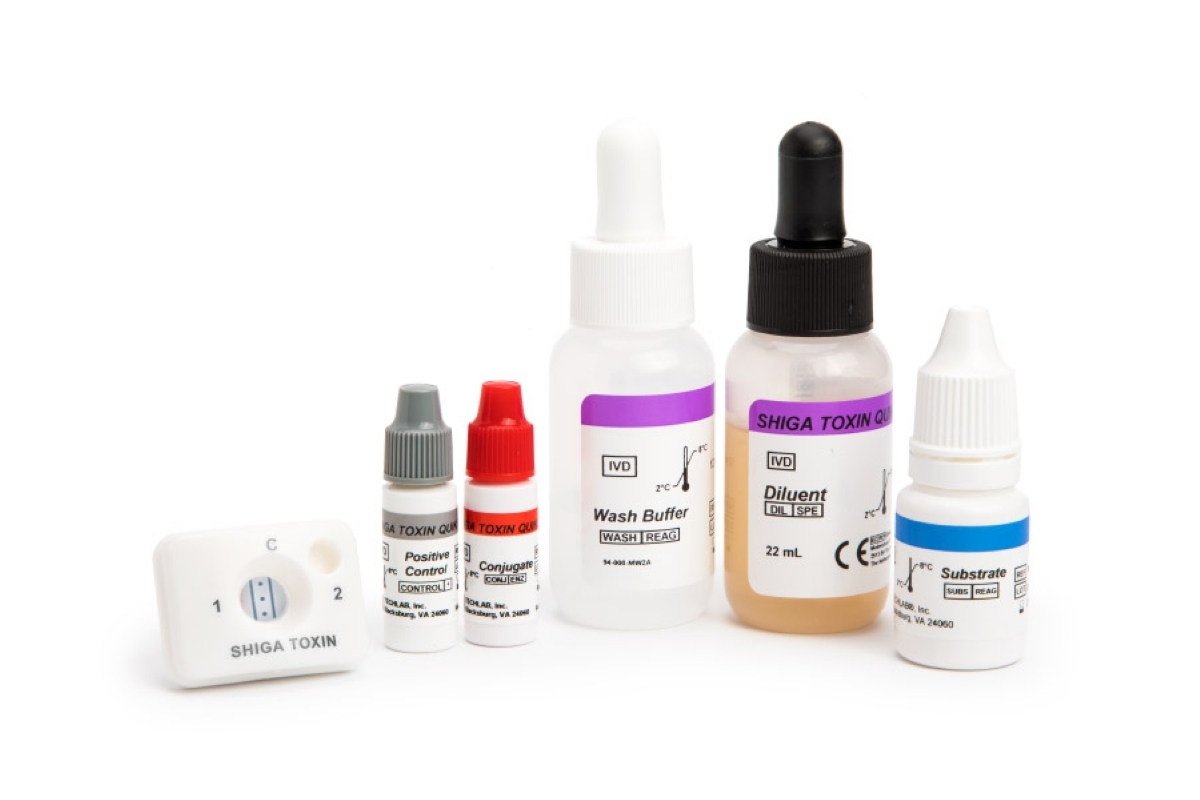
The SHIGA TOXIN QUIK CHEK ™ test is a rapid membrane enzyme immunoassay for the simultaneous qualitative detection and differentiation of Shiga toxin 1 (Stx1) and Shiga toxin 2 (Stx2) in a single test device.
It is intended for use with human fecal samples from patients with gastrointestinal symptoms to aid in the diagnosis of disease caused by Shiga toxin-producing Escherichia coli (STEC). It may be used with fecal specimens, or broth or plate cultures derived from fecal specimens.